
Objectives
- Evaluation argument for contamination control at the floor level
- Including the floor level in a holistic view of the contamination control process
The Story
Studies in the late 1960’s – 1970’s demonstrated that redistribution of bacteria into the air from the floor accounted for up to 15% of all airborne bacteria (Hambraeus et al. 1978) and disinfection alone is short lived given the rapid rate of recontamination on vast surface such as the floor (Ayliffe, 1967).
Current evidence surrounding floors as a key surface discusses how it may act as a reservoir in the chain of infection both in sensitive environments and potentially in other facilities as well.
The 2000’s started to paint a clearer picture of how floor surfaces acted as vectors of re-dispersion for microbes (Gupta, 2007). The evidence consistently points in the direction of floors playing a role in pathogen dissemination, especially dust particles (Prout, 2013). Backing earlier research linking the risk of human shedding issues (Hambraeus, 1978) & their pathogenic risk if airborne (Wei, 2016).
While there is limited research being developed on floor level controls outside of the healthcare setting, it is generalized as a relegated entity within a contamination control practice versus other surfaces.
The Science
The largest surface area in each room is seeing the least control across a variety of industries ….
Touch level and air surface cleanliness alone are not sufficient practices based on the criticality of the areas they are positioned in. At best, one of the most basic requirements in controlled environments, shoe covers, can themselves be a new vector of concern due to cross contamination between zones.
Define Vectors – A vector is a path for contamination to travel with minimum disruption or control through wheels, shoes and the surfaces as a whole
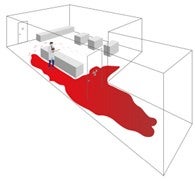
More people + more capacity + more activity = more particles + more risk = more control
Most end users spend 80% of budget/time/effort on 20% of a problem when a reversal can yield more sustainable, long-term gains.
Floor level control reduces substantial risk prior to controlled entry in a far more sustainable way.
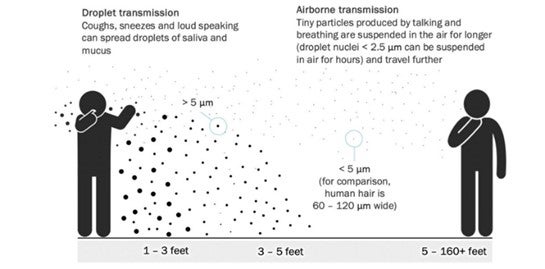
The ‘Sense’
Cases explored and the Onion.
The five second rule might apply in your kitchen, but it does not apply in sensitive manufacturing.
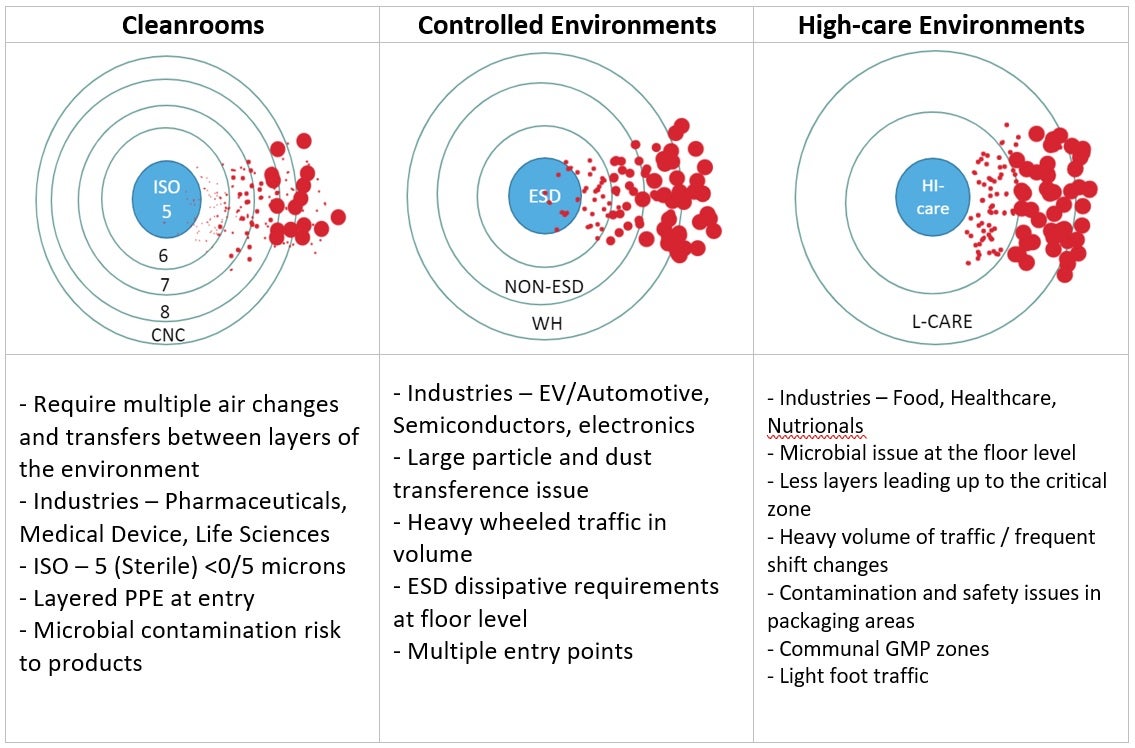
The following diagrams represent a concept presented by Dr. Tim Sandle, he calls it the onion concept. This concept illustrates the most critical area, the one you want to keep free of contamination at the center of the diagram. This could be a cleanroom, controlled environments, and/or high-care settings. The critical center area is protected by the outer layers (or other areas) that surround it. Certain measures are put in place in these outer layers to prevent contamination from entering the critical area, this can include hand washing, gowning and contamination control flooring. As personnel move through each layer, it becomes cleaner and cleaner until they reach the critical area in the center. Depending on the criticality of the site, it will determine how many layers or areas there are to move through before reaching the center.
You can see below a diagram of how particles at floor level may travel through each layer, the red particles show the general risk and most contamination comes from the outer layers inwards. The larger particles tend to be prevented earlier on and less particles are transferred as you move through the layers.
A selection of settings where this is applicable…
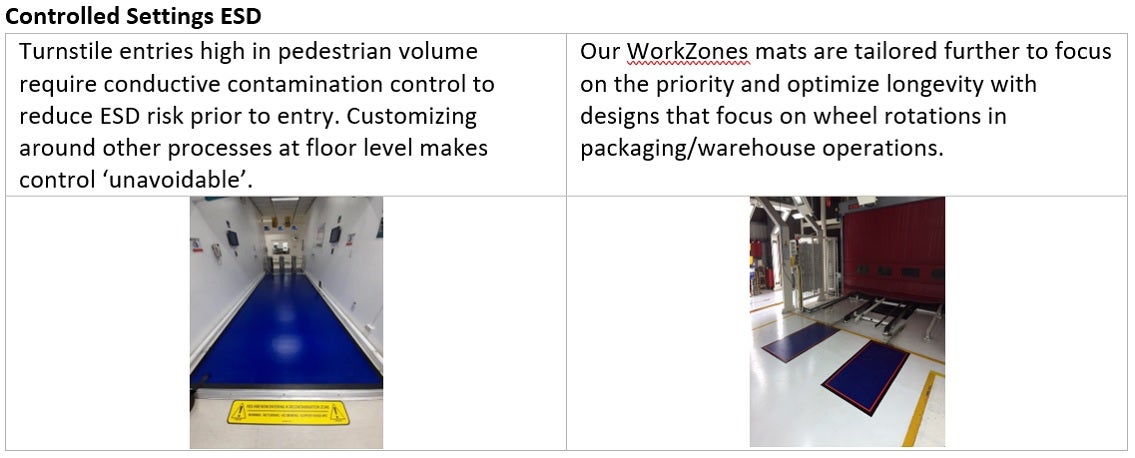
The Suggestion
Dycem Contamination control flooring alternatives.



The Statement
Moving forward, what influence will floors have on contamination control?
As Floors are a surface of high transmission, the contamination risk merits wider, more comprehensive research to value its part in a contamination control strategy (CCS). Part of this research considers the floor as a priority ‘vector surface’ to allow it to share equal weight in developing a CCS. If floors are treated for particulate control at the source of contamination, they can offer an exhaustive list of benefits for critical and controlled settings. Practitioners of CCS should approach the topic with a more holistic view to develop a CCS as part of their day-to-day, good manufacturing practices (GMP).
Dycem has vastly improved cross-contamination reduction through its ability to be unavoidable, simple, and a long-term, effective solution.
Posted by Dr. Tim Sandle, Pharmaceutical Microbiology Resources (http://www.pharmamicroresources.com/)

.jpeg)


